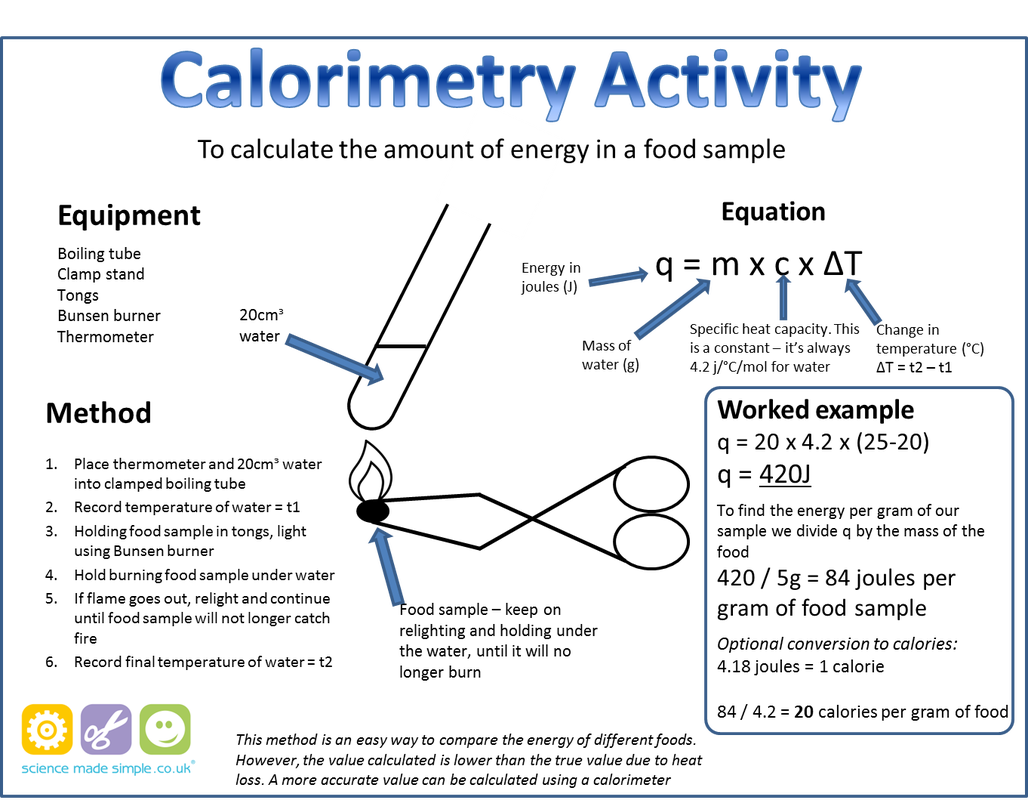
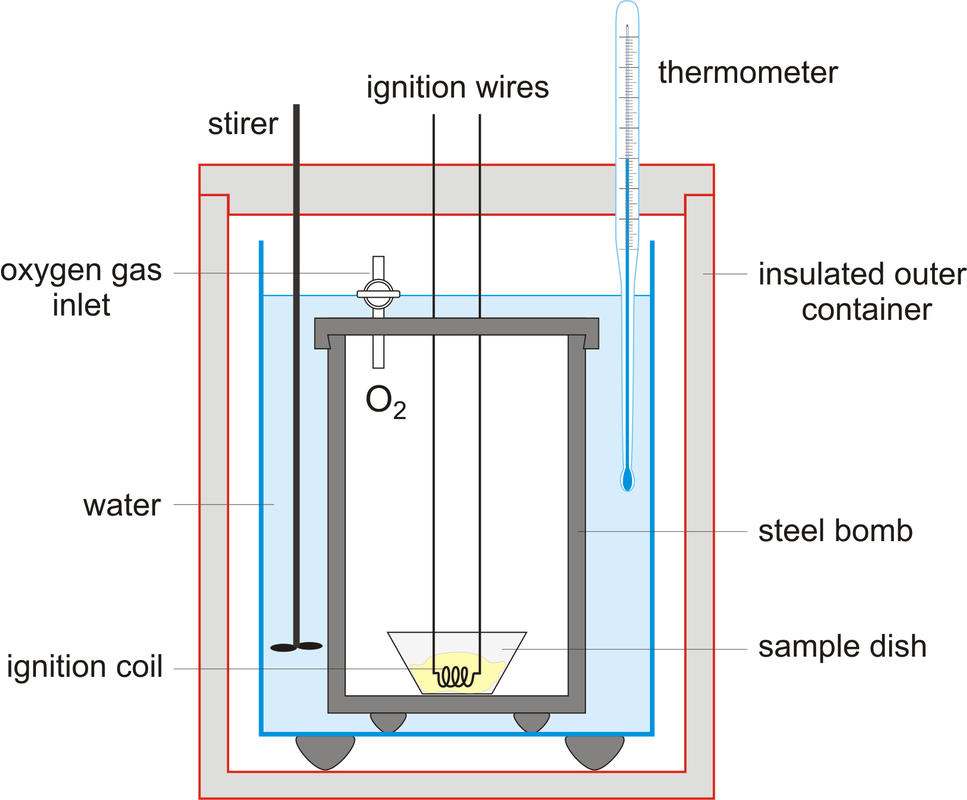
A calorimeter is used to measure the amount of energy in foods. The following features lead to more reliable results:
- The food is ignited electronically
- The food burns in oxygen and therefore burns completely without leaving a mass of carbon
- The combustion chamber is surrounded by water so little or no heat energy is lost from the system
- The stirrer leads to more efficient heat transfer and ensures that the water is of an even temperature.
| Respiration and photosynthesis for nat 5.pdf | |
| File Size: | 73 kb |
| File Type: | |
Rate Limiting Factors
|
If you want to print these concise notes, click Adobe file on the right to download as a pdf.
|
| ||||||
Your browser does not support viewing this document. Click here to download the document.
|
The more detailed notes shown above can be downloaded and printed by clicking on the file icon to the right:
|
| ||||||